
Clinical Rationale
Couple infertility affects approximately 15% of the reproductive-age population. Genetic causes account for a substantial proportion of cases (10–15% in male infertility; 8–10% in female infertility), while a meaningful share—around 20%—remains idiopathic even after a complete diagnostic work-up.1
In this context, having a single platform that integrates carrier screening and infertility genetics represents a major step forward, enabling optimized etiological assessment, stronger reproductive counseling, and more personalized assisted reproduction (ART) pathways.
1 Registro Nazionale PMA – Istituto Superiore di Sanità, Infertilità e tecniche di PMA, aggiornamento 09/03/2018.
Four Clinical Dimensions in a Single Analysis
CARRIERADVANCE Exome Infertility is a genetic test designed for couples with reproductive difficulties who are planning a pregnancy through ART.
The test integrates, in one analysis, four high-impact clinical areas:

Expanded Carrier Screening
Clinical Exome Sequencing:
- Clinical exome sequencing of 7,000+ genes associated with Mendelian disorders.
- Identification of the risk of transmitting genetic conditions to offspring, often in the absence of a family history.

Couple Infertility Genetics
- Diagnostic assessment of the main genetic causes of male and female infertility;
- Support in developing personalized clinical strategies.

TESE Prognosis in NOA
- Estimation of the likelihood of sperm retrieval in patients with non-obstructive azoospermia (NOA).
- Prognostic stratification TESE+/TESE− to support targeted clinical decisions.

Predisposition to Embryonic Aneuploidy
- Assessment of the risk of meiotic and post-zygotic mitotic errors.
- Useful in complex clinical scenarios (e.g., repeated implantation failure, recurrent pregnancy loss).
Technology Platform
CARRIERADVANCE
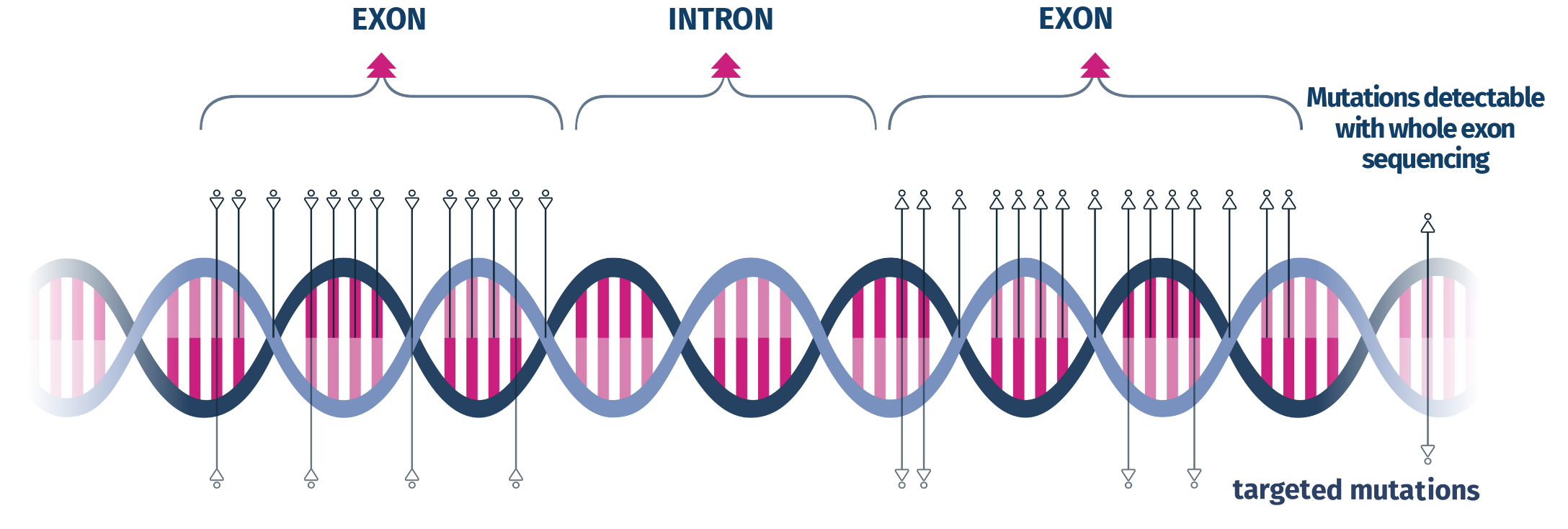
Exome Infertility is based on Clinical Exome Sequencing using state-of-the-art NGS technology, sequencing the entire coding region of ~7,000 genes. The integrated advanced bioinformatics pipeline enables detection of SNVs, indels, and CNVs in the investigated regions.
Advantage over targeted panels: no predefined mutation set; detection of any variant within the sequenced regions.
Report Interpretation
Carrier Screening & Infertility Genetics
POSITIVE: one or more variants identified (pathogenic or VUS).
Interpretation depends on the gene, inheritance mechanism, and clinical phenotype.
NEGATIVE: no relevant variants identified. Clinical decisions rely on comprehensive evaluation. Does not fully exclude genetic risk (the test does not cover the entire genome).
TESE Prognosis / Aneuploidy Predisposition
POSITIVE: pathogenic variant identified.
- NOA: contributes to TESE+/TESE− prognostic stratification.
- Aneuploidy: may indicate increased predisposition (to be integrated with clinical data and PGT-A results).
NEGATIVE: no variants detected in the analyzed genes.
Does not fully exclude genetic risk (the test does not cover the entire genome).
Indications for Testing
The test is recommended in the presence of:

ART pregnancy planning (preferred indication for the couple).

Idiopathic couple
infertility.

Donor conception: genetic matching of donor/partner.

Known or suspected family history of genetic disease.

Non-obstructive azoospermia (NOA): pre-TESE evaluation and post-negative TESE assessment.

Repeated implantation failures without apparent cause.

Recurrent miscarriage, particularly with evidence of embryonic aneuploidy.

High proportion of aneuploid embryos at PGT-A, especially if unexpected for maternal age.

PGT-A patterns suggestive of meiotic or mitotic errors (multiple aneuploidies, mosaicism).
A SIMPLE, STEP-BY-STEP WORKFLOW



Informational Brochure

CarrierAdvance Exome Infertility
Download brochureRequest information about CARRIERADVANCE
Fill in this form for a free consultation. One of our geneticists will contact you, free of charge and without obligation, to provide you with all the information you need.



 Italiano
Italiano English
English